Institut Charles Sadron News
Publié le 02/10/2020 par Krafft Marie-Pierre
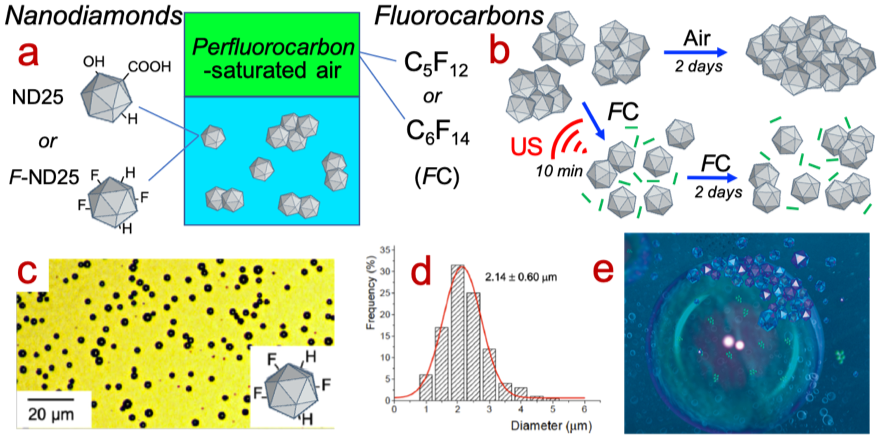
Despite their unique properties, the use of nanodiamonds in medicine is limited by their strong tendency to aggregate in aqueous solutions to form micrometric clusters. We show that the exposure of such aggregated nanodiamonds to fluorocarbon gases allows efficient disaggregation (Fig. 1a-b). This individualization enables fast diffusion of the nanodiamonds at the air/water interface, and thus, to generate for the first time, microbubbles with a shell made of nanodiamonds (Fig. 1c-d). The fluorocarbon effect is even stronger when the nanodiamonds are fluorinated, which unveils existence of fluorine-fluorine interactions that stabilize the interfacial film. The unique combination of the specific properties of fluorocarbons (hydrophobic and lipophobic character, low surface tensions, high vapor pressures with regards to molecular weight, etc.) and their chemical and biological inertness makes them highly attractive in medicine. Nanodiamond-shelled microbubbles have indeed a high potential for bimodal ultrasound/fluorescence imaging diagnostic and for the delivery of drugs and genes. The disaggregating effect induced by the fluorocarbons could generate novel applications, in particular in bioimaging and cell therapy.
This work has been achieved in collaboration with Marc Dubois (Institut de Chimie, Clermont Auvergne) for the synthesis of nanodiamonds, and published in ACS Applied Nano Materials.